Can a Scleroderma Registry Spur Clinical Breakthroughs?
A scleroderma registry launched more than 1 year ago is now compiling patient data and beginning to receive attention from clinical researchers seeking to study the complex, challenging disease.
Started by the Duke Division of Rheumatology and Immunology, the registry tracks disease presentations, symptoms, and responses to therapy. The registry includes patients from medical centers across the country.
“It has value as a repository for rare disease information and allows us to measure and monitor outcomes data,” says Ankoor Shah, MD, the rheumatologist who created the registry.
Shah follows nearly 100 patients in Duke’s scleroderma clinic. About one-quarter are included in the registry. “It is growing rapidly because patients with scleroderma are motivated, knowledgeable, and engaged in exploring treatments,” he says.
Patients in the registry are recruited and followed longitudinally in the secure Research Electronic Data Capture software to monitor their experience at Duke. The outcomes will be compared with those of other centers over several years.
Duke’s multidisciplinary scleroderma clinic attracts patients from a broad regional area because it offers convenient access to specialists who are knowledgeable about symptoms and new therapies.
Over the course of a few days, a patient can meet specialists trained to identify and treat the widely varied presentations of scleroderma. “The multidisciplinary aspect of our clinic is one of the most important advantages for a patient,” Shah says. “A patient can see a pulmonologist, dermatologist, gastroenterologist, physical therapist, and others as the condition requires.”
An orphan disease, scleroderma is often neglected in research because of limited commercial interest. But the Duke clinic and the scleroderma database are already attracting interest from researchers.
“The idea of a registry is attractive because clinical research organizations that are beginning research sometimes encounter trouble finding patients,” he says.
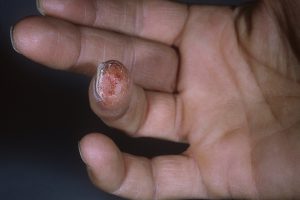
Two current trials, Shah says, illustrate the growing interest among researchers. Shah describes the trials as promising, but both remain in the analytical phase. A test of stem cells taken from a patient’s fatty tissue may help treat finger function and ulcers in Raynaud syndrome. A second trial is examining therapeutic options for lung disease, the leading cause of death among those with scleroderma.